Aurélie De Thonel and the V Mezger lab released a paper in Nature Communications elucidating the relationship between CBP and HSF2 in a neurodevelopmental disorder, CBP-HSF2 structural and functional interplay in Rubinstein-Taybi neurodevelopmental disorder.
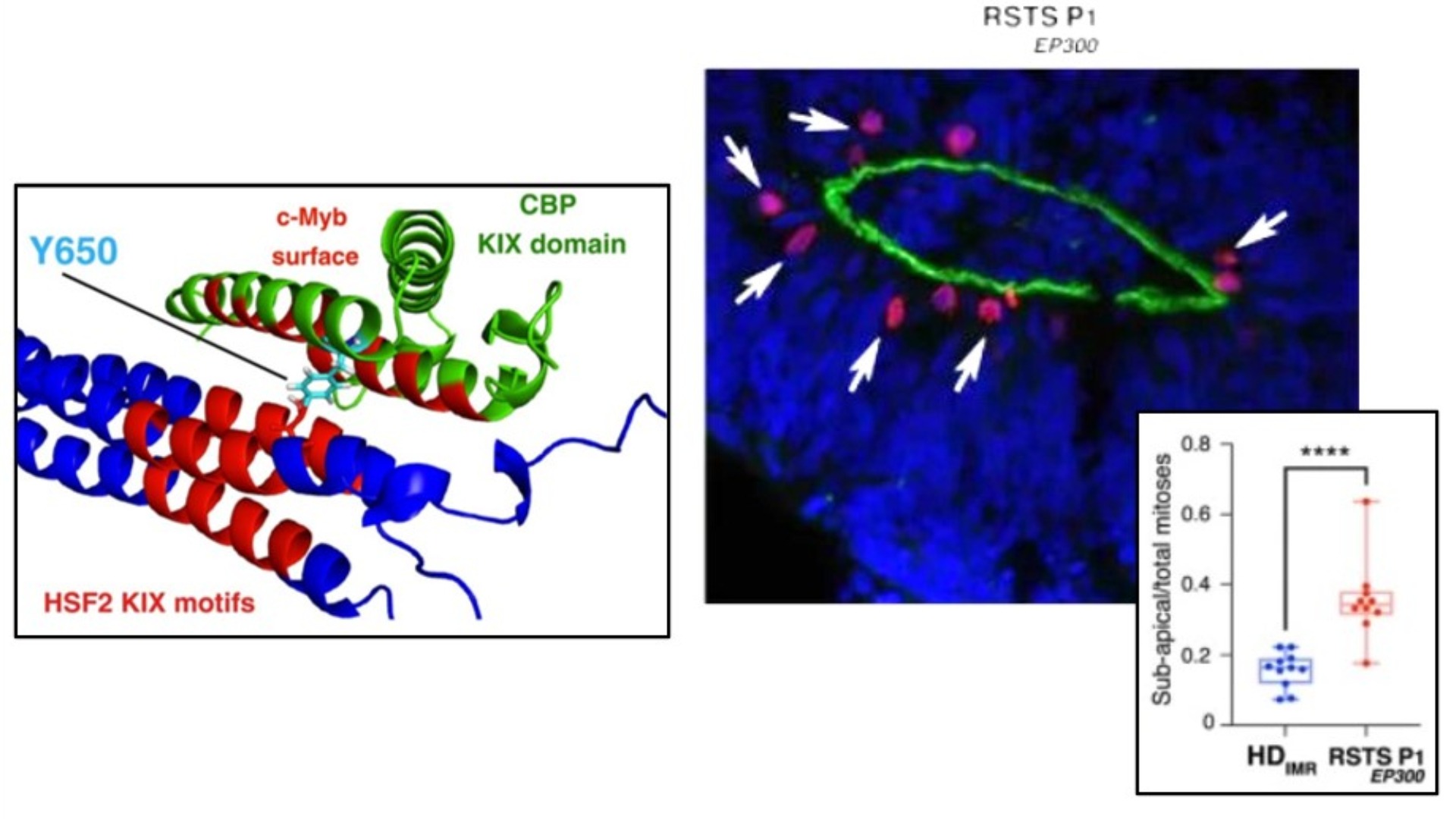
In the Rubinstein-Taybi syndrome (RSTS), patients carry dominant mutations in the histone/lysine acetyl transferases CBP or EP300 and suffer from neurodevelopmental disorder. The precise cellular dysfunctions causing the neurodevelopmental disorder remain however elusive as this enzyme have many targets beyond histone proteins. In this study, we show the contribution of a stress-responsive pathway in keeping the neural cell integrity as an early step toward the neural symptom of the disease. The heat-shock factor 2 (HSF2) is a fine tuner of brain cortical development and major player in prenatal stress responses in the neocortex. This study shows that CBP/EP300 acetylates HSF2, leading to the stabilization of the HSF2 protein. Consequently, RSTS patient cells showed decreased levels of HSF2 and their stress response was largely attenuated. Moreover, using 2D and 3D organoid models of cerebral development, the same CBP/EP300-HSF2 interactions were found as well as similar alterations of the stress response. Associated to this perturbed stress response, a decrease in a crucial neural cell adhesion protein, N-cadherin was observed and it could be rescued by stabilizing HSF2. Altogether the study highlights a crucial role of the HSF pathway in keeping neural cell integrity, a pathway particularly sensitive to stress and epigenetic alterations, two confounders of neurodevelopmental disorders.
Read more

Welcome to Orlane
Today, we welcome a new member to the team: Orlane. She's a student from the first year of the Brevet de Technicien Supérieur Biotechnologies en Recherche et Production (Two-year technical degree - Biotechnologies in Research and Production).Orlane will be working...

4th year PhD fellowships for Anaëlle
Congratulations to Anaëlle Azogui for obtaining a 4th year PhD fellowships from the "Fondation pour la recherche sur le cancer" (Fondation ARC). Read more

Congratulations to the Defossez team for their article published in Nature Communication
Congratulations to the Defossez team for their article “DNA methylation protects cancer cells against senescence” by Chen, Yamaguchi, et al., which has been accepted in Nature Communication. Read it here: doi:doi: https://doi.org/10.1101/2024.08.23.609297...

Welcome to Delphine, new post-doc in the team!
Delphine joins the lab as a post-doctoral fellow. She completed her PhD at the Centre de Recherche en Cancérologie de Lyon (CRCL) under the supervision of Virginie Petrilli. Her doctoral work focused on the pro-inflammatory protein NLRP3, for which she highlighted a...
